Chapter 3: Organic Chemistry
Organic Compounds- chemical compounds containing carbon
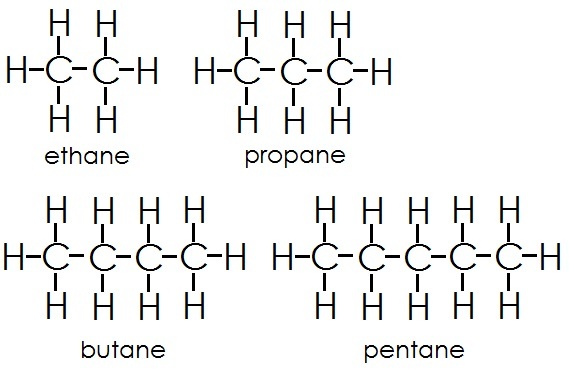
HydrocarbonsAn organic molecule consisting of only carbon and hydrogen
Carbon atoms can form many molecules because they have a valence of 4. The carbon atoms form organic compounds, which vary in shape and function depending on the functional groups attached to the compound.
|
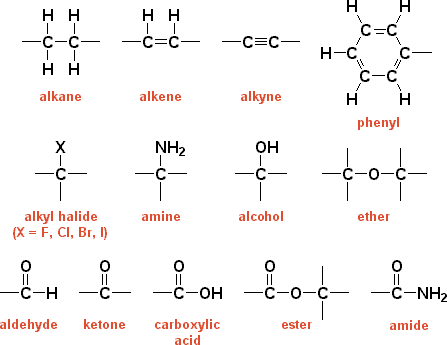
Functional GroupsA specific configuration of atoms commonly attached to the carbon skeletons of organic molecules and involved in chemical reactions
|
Macromolecules- a giant molecule formed by the joining of smaller molecules, usually by dehydration reaction. Polysaccharides, proteins, and nucleic acids are macromolecules.
monomer- the subunit that serves as the building block of a polymer
polymer- a long molecule consisting of many similar or identical monomers linked together by covalent bonds
polymer- a long molecule consisting of many similar or identical monomers linked together by covalent bonds
Synthesis and Breakdown of Polymersenzymes- a macromolecule serving as a catalyst, a chemical agent that increases the rate of reaction without being consumed by the reaction. most enzymes are proteins
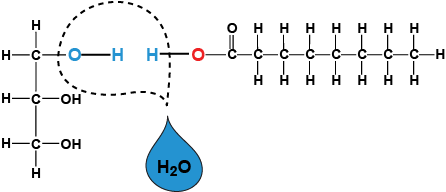
dehydration reaction- a chemical reaction in which two molecules become covalently bonded to each other with the removal of a water molecule hydrolysis- a chemical reaction that breaks bonds between two molecules by the addition of water; functions in disassembly of polymers to monomers Carbohydrates, proteins, and nucleic acids are all polymers, which are chains of monomers. Monomers join together through dehydration synthesis, or they can be broken apart by hydrolysis.
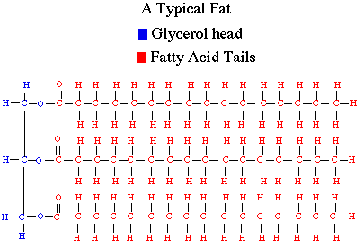
Lipidsany of a group of large biological molecules, including fats, phospholipids, and steroids, that mix poorly, if at all, with water
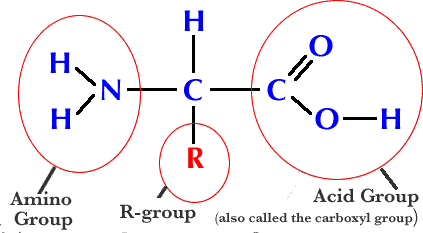
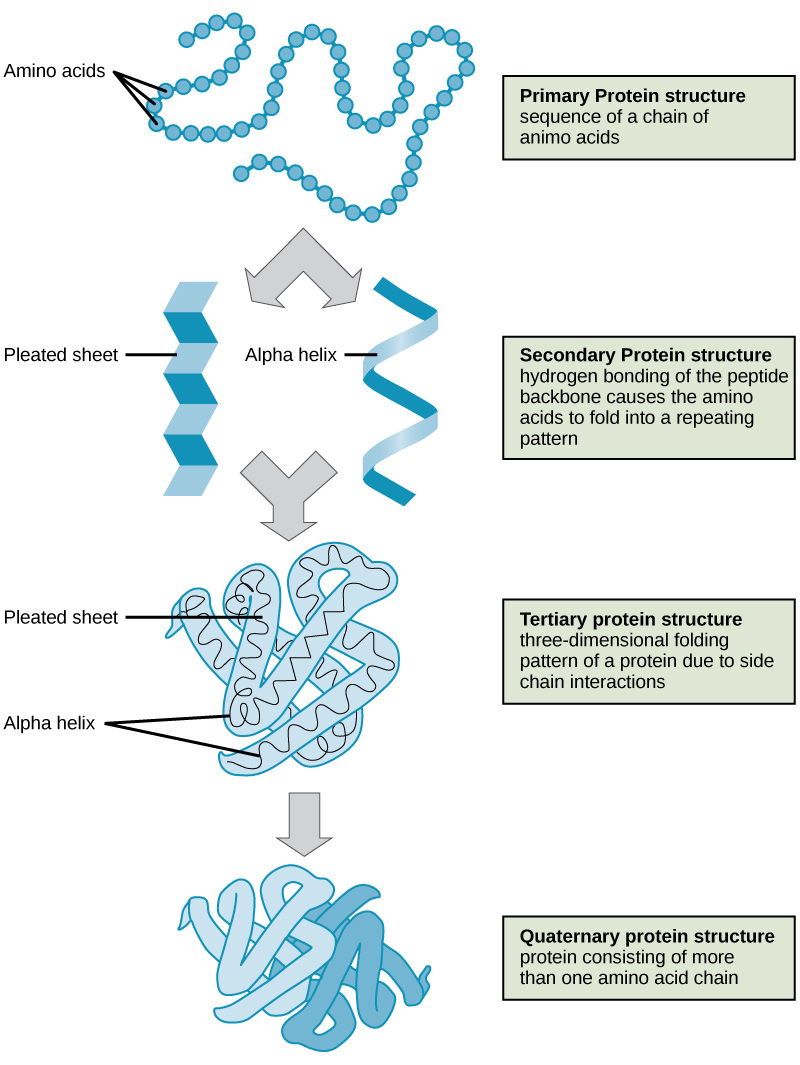
Glycerol bonds to three fatty acids through dehydration synthesis Proteinsa biologically functional molecule consisting of one or more polypeptides folded and coiled into a specific 3 dimensional structure
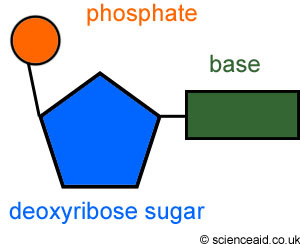
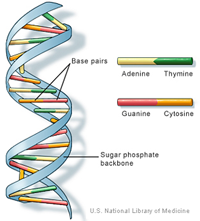
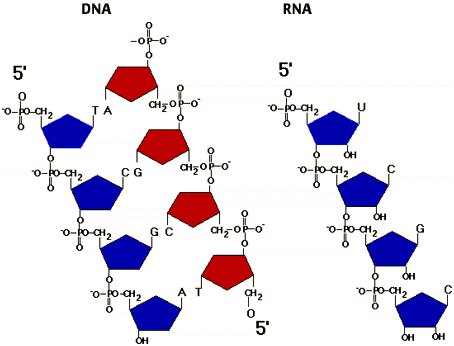
Nucleic AcidsThere are five different nitrogenous bases: Adenine, Thymine, Cytosine, Guanine, and Uracil.
The nitrogenous bases bond together with hydrogen bonds. Adenine bonds with Thymine or Uracil, and Cytosine bonds with Guanine. |
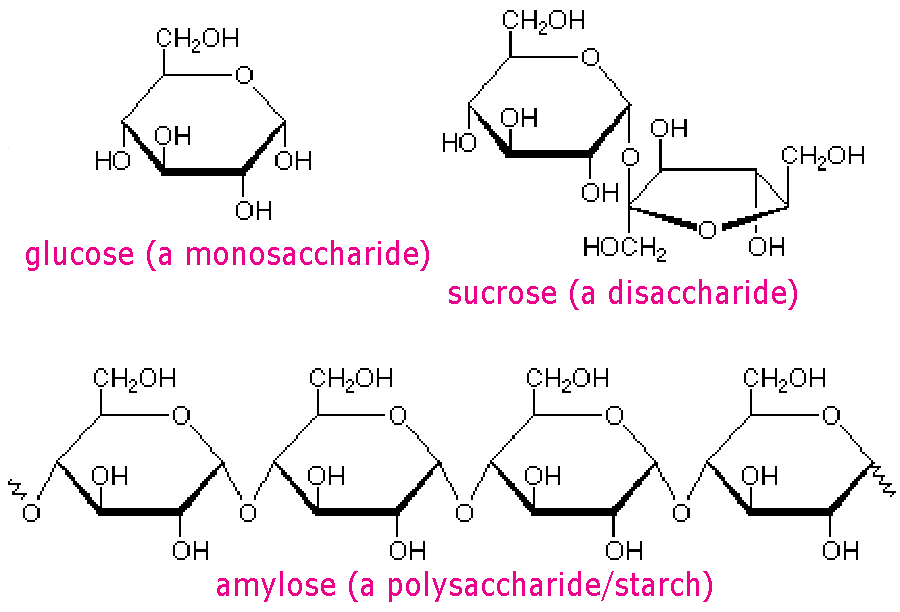
Carbohydratescarbohydrates- a sugar (monosaccharide) or one of its dimers (disaccharides) or polymers (polysaccharides)
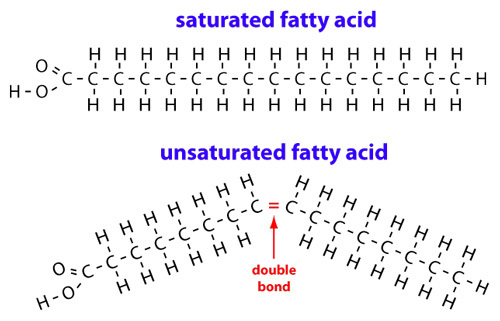
monosaccharides- the simplest carbohydrate, active alone or serving as a monomer for disaccharides and polysaccharides. also known as simple sugars, monosaccharides have molecular formulas that are generally some multiple of CH20 disaccharides- a double sugar, consisting of two monosaccharides joined by a glycosidic linkage formed by a dehydration reaction polysaccharides- a polymer of many monosaccharides, formed by dehydration reaction Types of Lipidssaturated fats- carbons have as many hydrogens as possible in the hydrocarbon chain; solid at room temperature
unsaturated fats- not as many hydrogens as possible in the hydrocarbon chain; double bonding between carbons; oils at room temperature Protein StructureDifferences Between DNA and RNADNA has A, T, C, G. RNA has A, U, C, G
DNA is a double helix. RNA is a single helix DNA has a deoxyribose sugar. RNA has a ribose sugar |